Paper: GS – II, Subject: Governance, Topic: Government Policies, Interventions, Issue: Eliminating HPV Cervical Cancer and Public Health Strategy.
Context:
India has launched a nationwide Human Papillomavirus vaccination (HPV) drive to prevent cervical cancer among adolescent girls. This is significant because cervical cancer is one of the most common yet preventable cancers affecting women in India.
Key Takeaways:
Background:
- Cervical cancer usually develops slowly through precancerous changes, which can be detected early through screening.
- In India, cervical cancer remains a major public health issue (2nd most common cancer in Indian Women after breast cancer) because many women are not screened regularly and diagnosis often happens at a later stage.
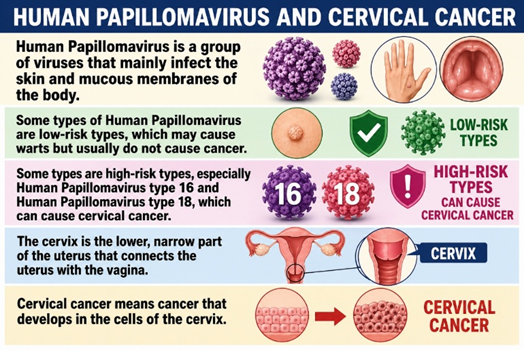
Explanation:
Target Group and Coverage:
- The vaccination drive targets adolescent girls, especially girls in the 9–14 years age group, before possible exposure to Human Papillomavirus.
- The vaccination is voluntary and free of cost.
- It will be provided through government health facilities such as Ayushman Arogya Mandirs and district hospitals.
Vaccine Used:
- India is using Gardasil, a quadrivalent vaccine.
- A quadrivalent vaccine means it protects against four types of Human Papillomavirus.
- Gardasil protects against Human Papillomavirus types 6, 11, 16 and 18.
- Human Papillomavirus types 16 and 18 are most important because they cause the majority of cervical cancer cases.
Dose Schedule:
- India has adopted a single-dose Human Papillomavirus vaccine regimen, based on World Health Organization recommendations.
- A delayed second dose may be given later, usually after 3 to 5 years, if required.
Safety and Effectiveness:
- The Human Papillomavirus vaccine is a non-live vaccine, meaning it cannot cause Human Papillomavirus infection.
- It works best when given before exposure to the virus.
- Studies show that Human Papillomavirus vaccines are highly effective in preventing cervical cancer caused by vaccine-covered Human Papillomavirus types.
Institutional Support:
- The vaccine has approval from the Central Drugs Standard Control Organisation, India’s national regulatory authority for drugs and vaccines.
- The vaccine introduction was recommended by the National Technical Advisory Group on Immunisation, India’s expert advisory body on vaccines.
- The programme is also supported through India’s partnership with Gavi, the Vaccine Alliance, an international organisation that helps improve vaccine access.
Campaign Strategy:
- The drive will be conducted as a special vaccination campaign, rather than only through routine immunisation.
- Coverage will be tracked through U-WIN, a digital platform for vaccination registration and monitoring.
WHO Elimination Targets:
The World Health Organization has set three major cervical cancer elimination targets by 2030:
- 90% of girls fully vaccinated against Human Papillomavirus by age 15.
- 70% of women screened with a high-performance test by ages 35 and 45.
- 90% of women with cervical disease receiving treatment and care.
Conclusion:
India’s Human Papillomavirus vaccination drive is a major step from cancer treatment to cancer prevention. Its success will depend on public awareness, vaccine acceptance, last-mile delivery, and regular cervical cancer screening.
Source: (The Hindu)
La Excellence IAS Academy, the best IAS coaching in Hyderabad, known for delivering quality content and conceptual clarity for UPSC 2026 preparation.
FOLLOW US ON:
◉ YouTube : https://www.youtube.com/@CivilsPrepTeam
◉ Facebook: https://www.facebook.com/LaExcellenceIAS
◉ Instagram: https://www.instagram.com/laexcellenceiasacademy/
GET IN TOUCH:
Contact us at info@laex.in, https://laex.in/contact-us/
or Call us @ +91 9052 29 2929, +91 9052 99 2929, +91 9154 24 2140
OUR BRANCHES:
Head Office: H No: 1-10-225A, Beside AEVA Fertility Center, Ashok Nagar Extension, VV Giri Nagar, Ashok Nagar, Hyderabad, 500020
Madhapur: Flat no: 301, survey no 58-60, Guttala begumpet Madhapur metro pillar: 1524, Rangareddy Hyderabad, Telangana 500081
Bangalore: Plot No: 99, 2nd floor, 80 Feet Road, Beside Poorvika Mobiles, Chandra Layout, Attiguppe, Near Vijaya Nagara, Bengaluru, 560040
HPV Vaccination India: Policy Shift to Prevention
